|
Tools
|
Proteins
|
Contacts
|
 |
Arabidopisis
seeds |
 |
|
Arabidopsis
thaliana, a small flowering annual dicotyledoneous plant, was
discovered by Johannes Thal (hence, thaliana) in the Harz mountains
in the sixteenth century. However, it is only since 1943 that Freidrich
Laibach reported for the first time the potential of this plant as
a model organism for genetic studies (Sommerville and Koornneef, 2002).
He was particularly interested in natural variation and the effects
of light quality and quantity on flowering time and seed dormancy.
One of the features of Arabidopsis that attracted him was the large
variation in physiological traits among accessions, which he started
to collect systematically in 1937. In 1943, he outlined the suitability
of Arabidopsis as a model for genetic and developmental biological
research because it produced large numbers of progeny and developed
rapidly, was easy to cultivate in limited space, had abundant natural
variation, produced fertile hybrids and had a relatively low chromosome
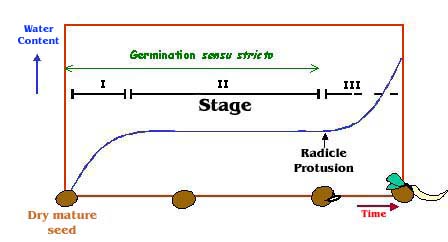
number (Laibach, 1943). Seed dormancy refers in seeds to the failure of a viable seed to germinate even when given favorable environmental conditions. In nature, dormancy mechanisms ensure that seeds will germinate at the proper time. Modern crops have been bred to exhibit reduced dormancy so that seeds can germinate immediately after sowing. Maturation drying is the normal terminal event in the vast majority of seeds, after which they pass into a metabolically quiescent state. Remarkably, seeds may remain in this state for many years, from decades to millennia, and still retain their viability (Hoekstra et al., 2001). Upon hydration under suitable conditions, the seed, if not dormant, reactivates its metabolism and commences germination, giving rise to a new plant (Bewley and Black, 1994; Bewley, 1997). Seed germination can be divided into three phases, imbibition, increased metabolic activity, and initiation of growth, which loosely parallel the triphasic water uptake of dry mature seeds (Figure 1). Morphologically, initiation of growth corresponds to radicle emergence; subsequent growth is generally defined as seedling growth. By definition, germination sensu stricto incorporates those events that start with the uptake of water by the quiescent dry seed and terminate with the protrusion of the radicle and the elongation of the embryonic axis (Bewley, 1997). Besides affording the appropriate physico-chemical conditions for reducing metabolic activity, the massive water loss occurring during late maturation is surmised to play a role in the switch in cellular activities from an exclusively developmental program to an exclusively germination/growth-oriented program (Kermode, 1990, 1995). Indeed, during seed development, metabolism is largely anabolic, being characterized by the synthesis and deposition of reserves. In contrast, during germination and initial plant growth, mobilization of the stored reserves occurs so as to provide an energy source for the growing seedling (Bewley and Black, 1994; Job et al., 1997; Eastmond and Graham, 2001; Gallardo et al., 2001). Despite numerous studies, the nature of the mechanisms involved in fine regulation of metabolic activity during maturation-drying and germination is still unclear. We are interested in determining the biochemical and genetic mechanisms that regulate the transition from quiescence to highly active metabolism during germination and seedling establishment. To this end, we develop a proteome analysis of the model plant Arabidopsis thaliana (Gallardo et al., 2001, 2002a,b). This global proteomics approach would allow characterizing the accumulation pattern of key metabolic enzymes in germinating seeds. mRNA profiling expression studies during Arabidopsis seed development (Girke et al., 2000) and barley seed germination (Potokina et al., 2002) have recently been reported. The data presented in this website are from our initial studies (Gallardo et al., 2001, 2002a,b). Newly identified proteins will be included progressively. |
 |
| References |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|